| Best View is Not in Email Please Click Here Now to Read Newsletter on VSI Website |
 |
||||||||||||||||||||||||||||||||||||||||||||
This Newsletter Has Been Generously Sponsored By: 
Click Above for More Information on Home Phototherapeutic Light Units for Vitiligo
|
||||||||||||||||||||||||||||||||||||||||||||
|

This is always an exciting time of year, as we attend the American Academy of Dermatology (AAD) Annual Meeting. This year’s meeting, held in San Francisco, CA from March 20-24, as in years past brought together the world’s foremost dermatology experts from all corners of the globe. I attended scientific sessions on treatment advances including exciting new discoveries about potential biologics. Development of such treatments is a direct result of ongoing research identifying new gene associations and pathways. Many of these discoveries are a direct result of our own MSAC member, Dr. Richard Spritz, and his NIH-sponsored genome-wide vitiligo study. This, and all, research would come to a complete standstill if it were not for the patient volunteers that sign up for the research studies. VSI members can give themselves a pat on the back for their continued support of, and participation in the many clinical trials VSI has announced in these newsletters. I have been especially interested over the past 2 or 3 years at the increase in not only awareness of, but concern for the patient’s point of view, and quality of life, as well as a growing acceptance of “patient-reported outcomes” in the research documentation. VSI’s input has been sought for a better understanding of the emotional impact of this disease. More physicians are recognizing the importance of patient-to-patient support, and referring their patients to support organizations like VSI. This is quite a departure from the old days when many, if not most, physicians held patient support organizations in contempt. In those days, having a patient seek outside information was seen by many as threatening and interpreted as a lack of confidence in the physician. I wish I could say that those days are completely behind us, but VSI still hears otherwise from enough patients to know better. I can, however, say with certainty that physicians of that type are on the decline and that the numbers of caring and compassionate physicians replacing them is most definitely on the rise. Medical education now addresses the importance of honest communication in developing a strong and trusting physician-patient relationship. This brings me back to the discussion of this year’s AAD meeting. I am always moved when sitting in a room filled with hundreds of young dermatologists and one of our vitiligo heroes, like Dr. Amit Pandya, Dr. Henry Lim, or Dr. Iltefat Hamzavi, becomes emotional during their presentation when telling the attendees that vitiligo is a disease of “bruised souls and bruised hearts” and that what patients want most of all - is just to know that you care. Not only are these men brilliant scientists, but these are the doctors who have come to VSI seeking the patient’s point of view, and have made it their mission to educate all young doctors on the importance of listening and caring. It is doctors like these that truly gives me hope for tomorrow.Sincerely, Why Does Vitiligo Remain in the Shadows? Vitiligo is one of the most prevalent autoimmune diseases, Yet - Vitiligo Remains…. If you look at the more successfully funded diseases, such as psoriasis,
But… One Thing VSI lacks the financial resources needed to strengthen the Together, We Can Make a Difference! Please Click Here Now to Show Your Support
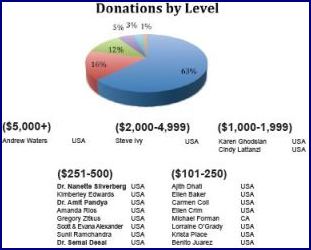
Donor Leaders to be Acknowledged! Donors who have responded to VSI’s Leadership Campaign
It’s Not Too Late to Join the Campaign! Please Show Your Support Today! * If you wish your gift to remain anonymous, please contact VSI and we’ll place your name accordingly.
Generic Drug War:
As most of our readers know, Protopic, a topical ointment approved by the FDA for eczema, is one of the most often prescribed (off-label) treatments for vitiligo. Because it is off-label, as well as quite expensive, many VSI members have experienced problems obtaining this prescription. In an effort to improve this situation, in 2012 VSI Executive Director Jackie Gardner met with the Vice President of Medical Affairs for Astellas, the manufacturer of Protopic. The two discussed the barriers that so many vitiligo patients encounter, and Gardner broached the possibility of adding vitiligo as an indication for the drug. Unfortunately, once a therapy is approved by the FDA for use for a specific disease, companies have little incentive to spend the millions of dollars it would take to enter new trials for vitiligo and get it approved for that as well. Another consideration affecting this type of decision would be how much longer the drug would be protected by its patent.
While the meeting did not achieve the desired outcome, VSI learned that Protopic’s patent was set to expire in little over 2 years (September 2014), and instead set its sights on the potential availability of the far less expensive generic form. Why are Generic Drugs Less Expensive? In America, due to the regulatory oversight of the FDA, a brand name drug can take 10-15 years of research and over 1 billion dollars to acquire the FDA approval to bring the drug to market. Free of the development costs that brand-name manufacturers incur, once the drug patent expires, generic drug manufacturers step in and make the generic copy for a fraction of the cost of the brand name drug, while still making a profit. This system worked well for many years, with very little regulation involved.
Protopic’s Patent Set to Expire in Late Fall of 2014 Looking forward to announcing this news, VSI kept a close eye on Protopic’s patent, which seemed inexplicably held up for several months. VSI Director Gardner held off filling her own prescription in the hopes of confirming the generic availability. Around mid-December, she submitted her Protopic prescription refill and was shocked at what followed. Her prescription had been written for a 100 gram (gr) tube. Her insurance company intervened and indicated that the prescription would need to be filled with the newly available generic drug, marketed as “tacrolimus.” The pharmacy had not yet received the generic product, and said as far as they could tell the 100 gr tube was not yet available from the manufacturer. However, the most startling news was the cost of this new generic. Because the 100 gr tube was not available, Gardner agreed to instead receive 3 of the 30 gr tubes of the tacrolimus ointment. Her cost after insurance for each 30 gram tube: $84.10. Her previous refill had been for a 60 gr tube of Protopic, (the name brand) with an “out-of-pocket” cost after insurance of $81.56.
Even more astounding was how little price difference there was between the name brand and the generic. It seems that when a generic drug is first released, due to the initial limited quantity, pricing can be just about as competitive as the market will bear. The prices shown below were obtained from a local pharmacy and are quoted at the full rate, without any type of insurance coverage.
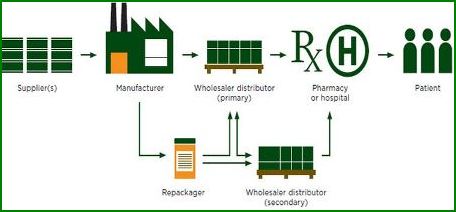
Though this generic was newly released, and in limited supply, a closer look soon began to reveal a far more pervasive situation with generic drug pricing. Piece by piece, VSI’s investigation began to uncover multiple layers of an apparent ongoing controversy regarding the skyrocketing prices of generic drugs, with increases in some cases as great as 8000 percent. A New York Times article from 2012, describing the rapid rise in generic drug prices as “mysterious,” focused on the ointments and creams that are the staple of dermatology treatments. One ointment, the topical steroid betamethasone dipropionate frequently prescribed for vitiligo patients, was quoted as jumping in price from $18.17 per tube up to $71.28. The article pointed out that the ordinary rules of supply and demand can be murky and illogical in the world of drug pricing. The influence and somewhat covert interplay of each of the components in the drug distribution chain adds to the over-all complexity. Once you figure in the insurance factor, which includes the all-powerful “Pharmacy Benefit Manager,” you truly have a mixing bowl of agendas rife for corruption. Drug Distribution Chain
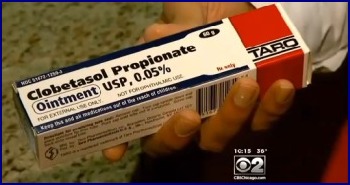
One doctor who also blogs on medical issues cites problems caused by cryptic deals made behind closed doors between the drug manufacturers and insurance companies, leaving the consumer with no idea of the true cost of a drug. In his September 2014 column titled “What is going on with generic drug prices?” he says, “It appears to me that the pharmaceutical companies have stopped their price competition, possibly by secretly dividing up the market and definitely by limiting supplies. If that is true, antitrust laws are likely being broken. Meanwhile, people with chronic illnesses are being squeezed financially even more than they already have been.” In a January 2015 interview with NBC Nightly News, Ralph Neas, President of the Generic Pharmaceutical Association (GPhA), denied greed as being a factor saying, ”there have been some increases, but if you put them in context you’re still basically talking pennies compared to very expensive brand medicines.” When asked about a drug jumping from 34 cents a day to $7.46, he replied, “it happens sometimes because there can be drug shortages, supply problems around the world, or a whole bunch of market forces.” Local/Community Pharmacies Also Suffer Collateral Damage Prices can go up dramatically practically overnight, leaving pharmacies at a loss when the pharmacy benefits managers (PBMs), who set the rates at which insurance companies reimburse the pharmacies, take weeks or months to update the rates. When this occurs, pharmacies are out hundreds of dollars when they are reimbursed far less than the cost they paid the drug companies for the drugs, as they base their prices on the out of date information, then find out weeks or months later that they will be reimbursed pennies on the dollar. Many local pharmacies are being driven out of business as they can't sustain the ongoing losses. Some pharmacists offered examples of the current situation. One mentioned that his pharmacy lost $3700 on a prescription (and multiple refills) they provided for a single patient, due to the lag in reimbursement rate adjustments. Others spoke of prices increasing over 300% several months ago, but the PBM continued to reimburse at the previous lower rate for over 6 months, resulting in losses of $40 to $100 or more per prescription that these generally small businesses can't absorb. Without these local pharmacies, patients are also profoundly impacted, as they have no place to go for their prescriptions, and also can't afford them. The National Community Pharmacists Association (NCPA) presented the information to Congress, requesting that bipartisan legislation be introduced to preserve patient access to pharmacies by requiring more timely reimbursement updates from PBMs. You can read more about this legislation below under the “Lawmakers Response” section. Could This Be A Double Whammy For Vitiligo Patients? Ominously, the problems with generics may be the beginning of an even bigger problem for vitiligo patients. Topical corticosteroids have long been the first-line treatment for many vitiligo patients, and insurance companies have historically covered the modest cost. In fact, even though side effects preclude continuous, long-term use, often required for treating vitiligo, insurers frequently require their use before they will prescribe a more expensive alternative such as Protopic. In October 2014, the CBS TV station affiliate in Chicago spoke with a local pharmacist on the soaring cost of generics. The pharmacist specifically cited clobetasol, a topical steroid frequently prescribed for vitiligo. He said that with no changes in ingredients or packaging, this product that used to sell for $10 jumped 3000% overnight to $300.00!
Who, or What, is to Blame? Though there is no clear indication of what’s behind this sharp climb in prices, there are enough theories to create a bestselling conspiracy novel. VSI’s research uncovered quite a bit of speculation and finger pointing. Read on to see what we found, and to learn more about the potential impact on vitiligo patients. Monopoly? Some cite monopoly as the culprit. But how could that be? From airline mergers to PC operating systems, the US government has in the past invoked its ‘anti-trust’ laws to protect its citizens from the unfair business practices such as “price fixing” that can result from business monopolies.
Gray Market Monopoly? Another type of monopoly is the “gray market monopoly.” Previously, once a drug patent expired and the generic drug manufacturers stepped in, this led to plenteous supplies and lower prices for patients. But now, with many of the generic manufacturers concentrating on the higher margins available in drugs newly available as generics, this leaves older, low margin drugs (like topical steroids) in the hands of a very few, or sometimes even a single generic supplier, creating what is known as a “gray market monopoly.” These monopolies are able to set prices at will since there is little to no competition, and shortages can be created when their production lines are unable to handle the demand, putting further upward pressure on prices. Greed?
Pharmacy Benefit Managers? Known as “drug plan middlemen,” the Pharmacy Benefit Managers (PBMs) negotiate drug prices from the manufacturers on behalf of their clients, such as pharmacies and private companies offering health benefits to large groups. According to In addition to the reimbursement practices cited above, other problems are attributed to a lack of transparency with regard to what is known as the “spread,” defined as the difference in price between what the PBM charges its clients for the prescription and the amount the PBM reimburses, or pays out to, the pharmacies that fill the prescription. In one example cited in a Fortune magazine article by Katherine Eban, a PBM billed a client hospital organization $92.53 for a prescription for generic amoxicillin, but only reimbursed the pharmacy $26.91. Many are calling for reform of these practices, suggesting new business models emphasizing transparency - requiring the PBM to disclose all costs, and to charge clients the same cost they paid, adding only an agreed upon administrative fee. ObamaCare?
Gray Market? Another interesting twist we uncovered is the shady area known as the “gray market.” Gray market companies, known to be unscrupulous, work outside normal distribution channels (shown in the graphic earlier in the article) to buy up all of the available supplies of targeted drugs at the low margin price, thereby artificially creating a market shortage, then turn around and sell the drugs through a variety of other companies for astronomical profits. In one example given by Rep. Elijah Cummings (D, Md.), a cancer drug was originally purchased from an authorized supplier for $7, then after making 9 stops at various points in the supply chain, increasing in price each time, was ultimately sold to a hospital for $600! Hospitals try to avoid dealing with these companies, but sometimes the scarce supplies make it impossible when a drug is vitally needed. “These companies are profiteers that zero in on short-supply drugs and sell them at inflated prices,” said Sen. Jay Rockefeller (D, W.Va.), chair of the Senate Committee on Commerce, Science and Transportation. Other Factors: Co-Pay Programs and Tier Creep As generic costs rise, insurance co-pays follow suit. What began as a way to help the consumer afford otherwise prohibitively expensive medications has now become a battle between drug manufacturers and insurers, with patients caught squarely in the middle.< “Tier creep” is driving up the costs of some medications. This happens when insurance companies decide to reclassify a drug from a lower tier to a higher tier with a resulting rise in the co-pay. For instance, says Chris Dowd, Senior Vice President for PSKW, a bio-pharma co-pay program, “Tier 2 drugs (medium co-pay) no longer carry a $25 copay, but a copay closer to $35–$75 — resulting in lower medication adherence.” A national study of more than 10 million prescriptions for some 5 million patients found that when co-pays reached $50, prescriptions were three times more likely to be abandoned at the pharmacy than when there was no co-pay, and more than twice as likely to be abandoned when the copay was $25.
Oh, What a Tangled Web We Weave……
What Does this Mean for Vitiligo Patients? What began as a good news announcement about a pricey vitiligo treatment becoming available as a more affordable and accessible generic drug instead raised new concerns. If the cost of something as simple as a topical corticosteroid can soar 3000% overnight, VSI is concerned that insurers may react by reclassifying their drug tier, resulting in higher co-pays, and making them as unaffordable as the higher priced treatments such as Protopic – or worse, deciding that since they, too, are off label - to deny coverage altogether. Lawmakers’ Response May, 2013: Rep. Elijah E. Cummings, Ranking Member of the House Committee on Oversight and Government Reform, introduced H.R. 5853, the "Gray Market Drug Reform and Transparency Act of 2012" to reform gray market drug sales. This Bill was referred to the Subcommittee on Health, but was not enacted. October 2014: Representative Cummings and Senator Bernard Sanders introduced legislation to cushion the impact on taxpayers when drug prices rise faster than the rate of inflation. In his testimony at a Senate hearing, Sen. Cummings was blunt in his assessment of the problem: “Some companies are exploiting monopolies and disruptions in supply to implement massive price increases in order to reap unconscionable profits.” Unfortunately, the Cummings/Sanders proposal was defeated; however, the problem now has the attention of many lawmakers. January 2015: Congress introduced H.R. 244 that will require the PBMs to adjust reimbursement rates (maximum allowable costs, or MACs) each week, which should help limit the losses to the pharmacies. The legislation also requires the PBMs to communicate the source on which they base their rate decisions, providing more transparency to the process. Hopefully, Congress will pass this bipartisan Bill! What Can YOU Do? You can write to your legislators and let them know how the high price of medications has affected your ability to obtain treatments.
Where Do Your Contributions Go? Every day VSI receives requests for information, support and services: from providing accurate and current treatment information, connecting patients with local doctors, to ensuring the voice and needs of the vitiligo patient are heard and understood. VSI’s capacity to serve depends on funding, manpower, and our ability to stay connected with the professional vitiligo community. Photos below were taken during the 2015 American Academy of Dermatology (AAD) Annual Meeting in San Francisco CA. This event provides VSI the opportunity to meet, exchange information, and network with vitiligo world leaders during a single meeting.  VSI Director Jackie Gardner, Vitiligo Researcher John Harris, &
recently appointed VSI Chairman of the Board Vicki Tiahrt
 Taken at the Coalition of Skin Diseases Booth in AAD Exhibit Hall
VSI Director Jackie Gardner, NIAMS Director Dr. Stephen Katz & Former Congressman Todd Tiahrt & his wife, VSI Board Chair Vicki Tiahrt
What's On Your Mind? Q. When using NB-UVB light treatments for vitiligo, how frequently should the light treatments be administered (i.e., once a week, twice a week, more frequently?)
Highlights of recently-published medical Measuring the Burden of Vitiligo As mentioned in the Director’s Message, in years past very little consideration was given to the patients’ point of view, and/or the emotional impact or burden of living with vitiligo. However, the realization over the past 2 -3 years that this aspect of the disease can play a critical role in overall prognosis and eventual treatment outcome, has resulted in emerging research validating the importance of addressing this aspect of the disease. The research outlined below has been developed specifically as a tool to validate and measure the burden of the disease. In 2013, two different scales were developed to measure the QoL in vitiligo patients – the VitiQoL (Vitiligo-related Quality of Life) and the Vitiligo Impact Scale. Both used similar methods in developing these scales, with slightly different results. The VitiQoL was developed by Lilly, et al. based on in-depth patient interviews and their responses to other dermatology-related indexes like the Skindex-16 and the Dermatology Life Quality Index (DLQI), and contains 16 items relating to life-participation limitations, stigma, and behavior. Shortly thereafter, Guarang, et al. published their Vitiligo Impact Scale, developed in similar fashion which also correlated well to the DLQI and Skindex-16 results. The objective is for these scales to be used to assess the psychosocial impacts of vitiligo in both clinical and research settings. In 2015, a French group including Dr. Khaled Ezzedine, and Dr. Alan Taieb of VSI’s Medical & Scientific Advisory Committee (MSAC) conducted a study to assess the burden of vitiligo in greater detail. They developed and validated a questionnaire to use as a tool to evaluate the burden of disease by focusing on skin phototype (coloring from fair to dark). Initially using a 35 item test on 300 patients, the final Vitiligo Impact Patient Scale (VIPs) consisted of 29 items. 21 items were common to both fair and dark-complected patients, while 3 were specific to fair skin, and 5 to dark skinned patients.
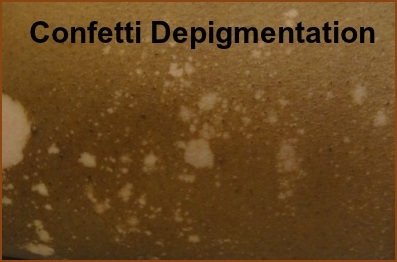
Their objective is for the test to be used to evaluate the burden of vitiligo in adults, Oxidative Stress and the Immune Two processes have long been thought to play a part in the development of vitiligo: oxidative stress and autoimmunity. Oxidative stress, an imbalance between oxidant and anti-oxidant levels in the body, can cause hydrogen peroxide to build up in the skin (among many other biological/physiological processes), damaging the melanocytes (pigment cells). Oxidative stress is the reason why research into using antioxidants either topically, in supplement form, or in the diet has increased. The relationship between autoimmunity and vitiligo has been well-documented, especially through the work of VSI MSAC member Dr. Richard Spritz, who has traced the genes involved in vitiligo and found many to be associated with several other autoimmune diseases like thyroid disease, autoimmune diabetes, and pernicious anemia. It is now generally accepted that autoimmunity plays a role in generalized vitiligo, though the exact processes remain under investigation. Until now, researchers thought that the oxidative stress and autoimmunity processes were separate. An Italian group, however, has just published a study (March 2015) that says that not only are both processes related, but they are synergistic. They theorize that perhaps the immune system creates a chronic inflammatory environment that allows reactive oxygen species (ROS – typically hydrogen peroxide) to proliferate and damage surrounding cells. The chronic damage that accumulates then triggers an immune response, creating a vicious circle of pigment damage. This publication cites another related study that analyzed anti-melanocyte antibodies (indicating autoimmunity) and lipid peroxidation levels (oxidative stress) in both recently developed vitiligo, and long-duration vitiligo. Interestingly, this group found that the lipid peroxidation levels were much higher in the recently developed vitiligo, while the antibodies were more prevalent in vitiligo of longer duration. They felt that oxidative stress might trigger the cycle of inflammation/damage/autoimmunity rather than the other way around. Either way, both studies point to the interrelationship between oxidative stress and autoimmunity in vitiligo. Confetti Depigmentation as a Sign of Rapidly Progressing Vitiligo Dr. Amit Pandya and his Dallas, TX research group are awaiting publication of his research into whether the presence of numerous (dozens to hundreds) small areas of depigmentation in a confetti-like pattern reveals rapidly progressing vitiligo in patients not receiving treatment.
They conducted a retrospective, single center chart review of 178 patients. 15 of those patients exhibited a confetti type depigmentation pattern, and 7 who met the criteria of having this pattern of depigmentation, a follow-up image available, and were not undergoing treatment, were ultimately included in the study. After evaluation of the 13 images from these patients that were assessed at baseline and follow-up 16 weeks later, they found that depigmentation had increased from 19% to 51% over that period, an increase of 32%! Two patients with an additional follow-up at 31 weeks had depigmentation that had increased to 71%, another 53% increase. They concluded that this pattern of depigmentation may indicate rapidly progressing vitiligo requiring immediate and aggressive treatment to stabilize the disease activity. Some dermatologists will treat aggressively advancing disease manifested by many new lesions, and widespread confetti depigmentation with oral systemic steroids. This type of therapy must be administered very carefully to avoid side effects and is typically prescribed with pulse dosing. For sources and additional information on this type of systemic therapy, see the newsletter Bibliography.
New Segmental Vitiligo Study in Detroit MI and Worcester MA
New Autoimmune Vitiligo Study in Detroit
New Needling Clinical Trial in New Jersey
Melanocyte-Keratinocyte Transplant Procedure
A Special Call To African-American Vitiligo Patients
NEW! - Support VSI Through Ebay
Earn Funding for VSI 3 Ways When You Shop! Please keep VSI in mind when you do any of your online shopping
|
|||||||||||||||||||||||||||||||||||||||||||
| Copyright © 2015 Vitiligo Support International Inc. All rights reserved. Reproduction or republication strictly prohibited without prior written permission A Vitiligo Support International, Inc. financial statement is available upon written request from the Virginia Office of Consumer Affairs. Mail requests to: Virginia Department of Agriculture and Consumer Services, Office of Consumer Affairs, P.O. Box 1163, Richmond, Virginia 23218. Click here to unsubscribe |
||||||||||||||||||||||||||||||||||||||||||||





 “Pharmacists United For Truth And Transparency,” “PBMs influence more than 80 percent of prescription drug coverage in the U.S. and administer drugs to more than 200 million Americans.” The three largest PBMs are Medco Health Solutions, Express Scripts and CVS Caremark.
“Pharmacists United For Truth And Transparency,” “PBMs influence more than 80 percent of prescription drug coverage in the U.S. and administer drugs to more than 200 million Americans.” The three largest PBMs are Medco Health Solutions, Express Scripts and CVS Caremark. 


 The dramatic spikes in generic prices have drawn the interest of multiple agencies, as well as the US Congress.
The dramatic spikes in generic prices have drawn the interest of multiple agencies, as well as the US Congress.