 |
|||||||||||||||||||||||||||
|

Dear Members and Friends of VSI, Vitiligo has been my companion for many years. While vitiligo is no longer the focus of my every thought, it still affects much of my daily routine and decisions. I am aware of the slightest changes in my skin. I tailor my diet, as well as supplements, around what may or may not be beneficial for vitiligo. I am acutely aware of times of increased emotional stress, and adjust my supplements accordingly in an effort to ward off a trigger of pigment loss. I gave my hairdresser a list of chemicals known to affect vitiligo. The lady who shampoos me knows to take care not to wash off the make-up at the top of my forehead. When I shop for clothing or shoes I give thought to styles that are more apt to cover depigmented areas. I take my treatment very seriously, carefully assessing the times I feel that I must aggressively treat, as well as those when I feel I can take a break. I tediously apply corrective make up (affectionately referred to as "war paint" by my husband) nearly every day of my life. Though I am comfortable with my life at the moment, I do think about the future. Will it spread? Will my children get it? When I grow old, will I still be covering it every day? For the first time since developing vitiligo, I can see the possibility of meaningful change during my lifetime, resulting in a more positive future International vitiligo meetings are now offering researchers unprecedented worldwide collaboration. By coming together and sharing ideas and resources, they overcome the challenges of isolation, and generate a global insight never before possible. 2012 marked a milestone in this process when an international group of world-renowned vitiligo scientists and clinicians recognized and declared that the missing piece of this research team was the vitiligo patient. Not only is the patient's history and point of view important - but without patient participation, research cannot continue. In each issue of this newsletter, we include new research information and opportunities for patient collaboration. I know that each and every one of you wants better treatments, and hopes for a cure, but wanting and hoping do not bring change. I ask that you demonstrate your passion and desire for change and a better future in two ways: Support research with your participation. Support VSI financially so that we can expand our Sincerely,
World Vitiligo Day 2013!
June 25, 2013 marked the second "World Vitiligo Day" (WVD). On this day, vitiligo groups all over the world held events dedicated to increasing a global awareness of this disease. I was honored to attend the USA World Vitiligo Day press conference and celebration held in Detroit, MI. The group included doctors, researchers, patients, and the heads of vitiligo organizations, and support groups from across the world. The common bond we shared created such a feeling of camaraderie. This is just the first step of a political collaboration to raise our voice and be heard as a community.
Vitiligo Treatments: How Do They Work? Vitiligo is an autoimmune pigmentation disorder that destroys the melanocytes (cells which make pigment) leaving skin and hair depigmented. Melanocytes in vitiligo patients, whether in normal or depigmented skin, are inherently different from those in people without vitiligo. This “difference” may make them uniquely vulnerable. What triggers and drives their destruction? There are several theories as to what is triggering and influencing this autoimmune dysfunction. Many believe that this likely means that vitiligo is the result of a multi-faceted set of factors, including a complex set of genes, stress, accumulation of toxic compounds, infection, autoimmunity mutations, and impaired melanocytes. Research is focused on understanding this interplay of factors in order to improve existing therapies and, for the first time, be able to design therapies to stop depigmentation. Current vitiligo therapies such as topical steroids, topical immunomodulators (non-steroidal alternatives that affect the function of the immune system without the side effects associated with topical steroids), excimer laser, narrowband UVB (NB-UVB), and psoralen plus UVA (PUVA), are aimed at suppressing the immune response so that the melanocytes once again flourish and repigment the skin. These therapies are used according to the type and severity of the vitiligo. For example, topical and laser therapies would typically be used primarily to treat smaller, isolated lesions of vitiligo, whereas full body NB-UVB or PUVA would be used to treat more widespread vitiligo. Though these conventional therapies were initially developed for other skin diseases, they are also moderately effective in vitiligo. By suppressing the immune system’s dysfunction and stimulating melanocyte growth, these treatments can help the skin regain its pigment. In recent years, various topicals, such as topical immunomodulators (tacrolimus or pimecrolimus), have been successfully combined with NB-UVB to shorten treatment time and speed up repigmentation. Other combinations are now being tested, as well as adaptations of other conventional drugs.
In addition to improving our ability to suppress the immune response through adaptations of existing therapies, we may eventually have the capacity to interrupt the immune response that results in vitiligo. Several researchers are looking at ways to interrupt the biochemical signaling that leads to the depigmentation of the skin. The object is to prevent depigmentation by interrupting the cascade of biological events leading to the destruction of melanocytes. The progression of existing vitiligo would be stopped and depigmented skin would be able to naturally repigment. Normal messaging would be restored, thereby enabling the body to once again produce the healthy melanocytes needed for pigmentation. If successful, this research could lead to more effective and safer methods of control, and constitute the first therapies uniquely designed to treat or prevent vitiligo. Adaptations of Existing Therapies (Repurposing)
Vitiligo researchers continue to look at a range of already-approved therapies for their applicability to vitiligo. One such example would be etanercept (Enbrel), which was found to be an effective therapy for several autoimmune diseases through its ability to inhibit the tumor necrosis factor (TNF) which contributes to the inflammatory process that causes many of the problems in autoimmune diseases. As high levels of TNF have been observed in vitiligo patients, researchers speculated that etanercept might also be effective for vitiligo. Ultimately, etanercept was found not to be effective in vitiligo. Tests of two other anti-TNF agents, infliximab and adalimumab, were also unsuccessful for vitiligo. Other therapies, however, offer promise. Below we review these therapies and where the research stands. In at least one case, there is an opportunity for vitiligo patients to sign up through our newsletter to participate in a clinical drug trial. We begin with afamelanotide, a drug being tested in combination with NB-UVB. Afamelanotide
Afamelanotide is a man-made chemical analogue (similar, but not exactly the same) of a naturally occurring hormone called alpha-melanocyte stimulating hormone (α-MSH). In nature, α-MSH is our body’s response to skin damage from ultraviolet (UV) light. This hormone activates melanocytes to make pigment. Afamelanotide mimics the effects of the natural-occurring α-MSH, but also has a more long-lasting effect and greater potency. Photocil In addition, Photocil may prove to be an important adjunct to traditional phototherapy delivered in a clinic. Some loss of pigment occurs in many patients within 1-2 years after successful treatment at a phototherapy clinic. Daily Photocil use might prevent or minimize the pigment loss. A future study will need to be conducted to evaluate the use of Photocil as a maintenance therapy. Red Light Therapy Visible red light has been shown to stimulate melanocyte growth, NASA discovered over forty years ago that red light stimulates plant growth. It was also found to promote cell repair and healing of injuries incurred by astronauts while in space. NASA discovered over forty years ago that red light stimulates plant growth. It was also found to promote cell repair and healing of injuries incurred by astronauts while in space. Long-wave light is weaker than short-wave light, and therefore causes less skin damage. Because red light is long-wave, it is easier on the skin than the short-wave (NB-UVB, or UVA light in PUVA) light more commonly used to treat skin diseases such as psoriasis and vitiligo. Dr. Mohammed AlJasser, co-investigator of the red light therapy study, told VSI: "the trial we are currently conducting using red light is based on studies done by a Taiwanese group." In that study, 30 patients with segmental-type vitiligo on the head and/or neck were enrolled in the study. Red helium-neon (He-Ne) laser light was administered locally, once or twice weekly. After an average of 16 treatment sessions, initial repigmentation was noticed. Marked repigmentation was observed in 60% of patients with successive treatments. To see if you qualify for this vitiligo red light clinical trial, please see the study listed below in the Research and Clinical Trials section of this newsletter.
Sodium oxodihydroacridinylacetate (ODHAA)
The serum interleukin 6 (IL-6) level prior to treatment was found to be a good predictor of success in stopping the disease’s progression. Elevated levels of IL-6, a pro-inflammatory cytokine, have been found in vitiligo patients and are believed to play a role in melanocyte destruction and the spread of vitiligo. IL6 is a protein that is associated with inflammation and the production of B cells, a type of lymphocyte (subset of white blood cell) that attacks foreign substances in the blood. Researchers in India will be reporting later this year on their work showing that increased levels of IL6 are linked to active vitiligo. In their study of 60 active and 20 stable vitiligo patients, 25 UVB-treated treated patients, and 70 healthy patients, they found significantly increased levels of IL6 in the vitiligo patients, especially in the active vitiligo patients. This IL6 link is yet another autoimmune flag. ODHAA, called Neovir, is an inexpensive, non-prescription immunomodulator that is widely available in Russia. The investigators said it was not known if and when this drug would be available in the United States or in Europe. Simvastatin The Canadian doctors writing up this experience in a medical journal noted that simvastatin has been reported to have immunosuppressive effects and that its immune-modulating properties may be suppressing the hyper-immune activity responsible for the depigmentation in vitiligo. Statin therapy is known to down-regulate inflammatory activities in the inner lining of arteries that collect plaque. As part of its activity, simvastatin has been found, among other things, to suppress tumor necrosis factor-α (TNF-α) and its production of IL-18. Elevated levels of TNF-α have been found in the keratinocytes of vitiligo patients and have been linked to the destruction of melanocytes. Simvastatin’s suppressive effects on inflammation and TNF-α may interrupt the depigmentation process. Continuing Research into Existing Vitiligo Therapies There also are researchers looking at treatment approaches not focused on autoimmunity. A German group headed by Dr. K. Schallreuter developed a cream called pseudocatalase, that they report works as an antioxidant in relieving the oxidative stress they observed in vitiligo. Oxidative stress occurs when there is an accumulation of toxic compounds that are damaging the body’s cells. One of these toxic compounds, hydrogen peroxide, is especially destructive to the melanocytes. Hydrogen peroxide, a routine but toxic by-product of normal metabolic processes, can be found in everyone's skin; however, those with vitiligo tend to have higher levels. The body produces an enzyme called catalase that breaks down and controls the hydrogen peroxide, and protects the cells from the oxidative stress build-up. However, those with vitiligo do not produce enough catalase, which can result in much higher levels of the hydrogen peroxide. Dr. Schallreuter's unique pseudocatalase formula, when combined with light therapy, helps the skin break down the hydrogen peroxide, thereby lowering the oxidative stress. Dr. Schallreuter first reported on the efficacy of pseudocatalase in repigmenting vitiligo-affected skin in 1995. Though many researchers feel that oxidative stress may play a role in vitiligo, since they are unable to replicate Dr. Schallreuter's pseudocatalase formula, others have not been able to reproduce her results. Dr. Schallreuter’s team has continued their work with this cream and, most recently, reported that it may also be effective against hair graying. According to recent scientific studies, the imbalance of catalase and hydrogen peroxide may also play a role in causing gray hair. New Discoveries May Lead to New Therapeutic Approaches Through basic research, we are learning what proteins and pathways are involved in driving the appearance of vitiligo. We know that the immune system is overreacting to an unknown trigger, thereby producing too many T cells (the other type of lymphocyte that normally goes into cells affected by a virus or tumor) that are attacking healthy melanocytes. We are steadily learning about the pathways involved in this immune response, as well as the various environmental factors that may be triggering this response. With this information, scientists are now working on designing therapies aimed at interrupting the pathway involved in the expression of vitiligo. These new therapies would be the first uniquely designed to treat, or even prevent vitiligo. One of these potentially new designer vitiligo therapies that has been recently publicized is based on the research being done by Dr. Caroline LePoole and her colleagues at Loyola University’s Stritch School of Medicine in Chicago. They have created a modified protein that appears to halt the autoimmune response that causes pigment loss in vitiligo. They introduced the modified version of the protein, HSP70i, known to be involved in the autoimmune response in vitiligo, into two groups of dark colored mice that typically develop white patches of hair associated with vitiligo. The first group consisted of mice that develop these patches at 6-9 months old. When this group was injected with HSP70i several months prior to the time they would normally depigment, none of the mice depigmented.
Photos courtesy of Loyola University Chicago Stritch School of Medicine The other group, mice that rapidly develop white patches, was treated at the point when they'd already lost most of the pigment cells from their skin. Within a matter of weeks, the majority regained their pigment, growing back dark hair instead of white. They concluded that this modified protein’s success in mice strongly suggests it has great value for treating vitiligo in humans. Though we have many questions yet to answer about vitiligo, we have learned that vitiligo is not a single disease that lends itself to one treatment or permanent method of control. It is a disease of complex factors that affect what treatments will work for what patient. For example, segmental vitiligo (SV) and nonsegmental vitiligo (NSV), the two primary forms of vitiligo, may differ more in their cause and symptoms than previously thought, which is why they respond differently to treatments. SV may not have the autoimmune links of NSV, which may explain why vitiligo treatments that suppress the immune response don’t work well in SV patients. Published findings continue to underscore NSV’s strong links to autoimmunity, including NSV symptoms such as itching prior to depigmentation; a family history of prematurely graying hair; and emotional stress as a trigger, as well as elevated levels of various immune-related T cells and cytokines. Through the work of Dr. Spritz and his international team, we now know there is a genetic component to vitiligo. Multiple vitiligo genes have been identified, some linked to the melanocyte, others to immune system regulation, and some identifying association with other autoimmune disorders.
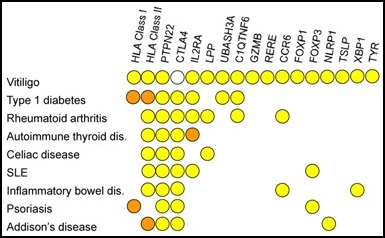
Genetic relationships of generalized vitiligo susceptibility genes and other autoimmune diseases.
While Dr. Spritz and others are identifying the genes that make the body susceptible to vitiligo, others are looking at the vitiligo lesion itself and the biological/chemical pathway to understand what is happening in the body during the vitiligo disease process. This research will make it possible for therapies designed to treat or prevent vitiligo to become a reality. Research Momentum Depends on Collaboration Going forward, researchers will be working to identify additional genes linked to vitiligo; to understand the sequence of events in the autoimmune dysfunction in vitiligo and the role of oxidative stress; and to describe the differences between SV and NSV and the causes and characteristics of the various subtypes of vitiligo within these primary forms. Their success will continue to depend on the willingness of vitiligo patients to participate in clinical trials, and share their personal histories. Without this, research will be stalled. VSI patient volunteers have been crucial to the current level of research progress, and we thank all of you who have participated to date. Dr. Spritz recently made an urgent request for everyone who has previously participated in his survey to contact him to update his or her information. He is particularly interested in treatment history and outcomes. Dr. Spritz is hopeful he can use the existing genetic data to better understand which treatments work or don’t work in different people and why. Gene research and patient experience together are not only informative, but also crucial to finding the triggers and causes of vitiligo, as well as to developing new treatments. Our understanding of how the disease behaves, both physically and biologically, and whom it affects is critical to developing effective therapies. Vitiligo research is moving at an unprecedented pace, but your support and participation is a critical part of this equation. Be sure to read the "Research and Clinical Trials" section of this newsletter, and on VSI's website regularly, to check for your opportunity to make a difference.
A Personal Message From Dr. Richard Spritz
What's On Your Mind? Q. Is my premature graying hair connected to my vitiligo?
Q. Is there any risk involved in waiting a while before treating my vitiligo?
Q. Why does my face repigment faster and better
Highlights of recently-published medical Early treatment is found beneficial for segmental vitiligo patients Korean investigators report that, contrary to common belief that segmental vitiligo (SV) is not responsive to conventional treatments, they found that SV was indeed responsive to therapy if treated early. They cite previous reports showing that SV responded poorly to phototherapy compared with non-SV, but they point out that the duration of disease was not looked at as a factor in those studies. In a retrospective study of 39 patients, they found that those with SV for less than five months (Group 1) responded much better to treatment than those having SV longer than 5 months (Group 2). Half of the 20 patients in Group 1 experienced more than 50% repigmentation as opposed to 26% in Group 2. The investigators found that treatments such as oral steroids, topical steroids, topical immunomodulators like tacrolimus and pimecrolimus, and phototherapy were effective when used early after onset of SV. The authors point out that these findings confirm the observations reported in two other 2011 studies that also showed that disease duration affected treatment outcomes in SV patients. Editor’s Note:The benefits of early treatment have also been found in NSV patients. In a 2012 Iranian study of 63 patients with generalized vitiligo, patients with recent vitiligo responded better to treatment than those with long-standing disease. For those with recent vitiligo (four years or less), the mean clinical response rate was 51.94% as compared to 40.04% for long-standing vitiligo (over four years). Also, over 61% of those in the recent vitiligo group had a “good or excellent response” compared to 27% in the long-standing group.
Autoimmune thyroid disease is linked to a more widespread vitiligo Autoimmune thyroid disease (AITD), especially Hashimoto’s thyroiditis, is a risk for vitiligo patients, with vitiligo generally appearing before or around the same time of the onset of thyroid disease. Two new large-scale studies now confirm that vitiligo patients who have AITD may have more vitiligo body surface involvement than those without thyroid disease. One study of 700 vitiligo patients was done by Belgian researchers and another by a French team studying 626 vitiligo patients. Both studies found that the percentage of total body surface involvement was significantly higher in those also having AITD, and greater in women when compared with men. Other variables identified in vitiligo patients as being associated with developing AITD included stress as an onset factor, a family history of AITD, and the duration of the disease, specifically noting that the risk of AITD doubled every 5 years after a vitiligo diagnosis. Recommendations from both groups are that NSV patients experiencing disease progression and/or with greater body surface involvement are more likely to develop AITD and should be monitored for thyroid function and antithyroid antibodies on a regular basis. Editor's Note: The thyroid hormones contribute to a healthy metabolism rate, growth, and cognitive function; clinical hypothyroidism negatively affects these processes. We do not know if subclinical (without symptoms) thyroid disease has negative consequences, but routine screening is recommended.
Severity and location of vitiligo Certain autoimmune/autoinflammatory diseases, especially autoimmune thyroid disease (AITD), seem to occur with greater frequency with non-segmental vitiligo (NSV) than in the population at large. Researchers at Ghent University Hospital in Belgium reported that these autoimmune diseases appear to influence the clinical characteristics (location and extent) of vitiligo. From their evaluation of 700 patients with generalized vitiligo, including 108 patients with associated autoimmune diseases, they have concluded that those with AITD have distinctly different clinical characteristics that could help predict the course of vitiligo. In vitiligo patients that were autoimmune/autoinflammatory disease-positive, the Belgian researchers found that the vitiligo tended to be more extensive, most often affecting the face, followed by the hands, then the extremities. The researchers speculated that, if patients do not have vitiligo on their extremities, especially on their hands, they most likely do not have thyroid disease. Conversely, if patients have depigmentation on their hands, wrists, ankles and elbows, they may be good candidates for screening for thyroid abnormalities. Editor's Note: Thyroid disease was the most frequent associated autoimmune disease found in the vitiligo study patients. Psoriasis, diabetes, and alopecia areata were the next most frequent, though to a much lesser degree.
Clinical Trial in Canada
Southern California Research Clinical Study
Clinical Trial in Massachusetts!
Your Opportunity to Move Research Forward!
Patients and Controls Invited to
Earn Funding for VSI 3 Ways When You Shop! Please keep VSI in mind when you do any of your online shopping
|
||||||||||||||||||||||||||
| Copyright © 2013 Vitiligo Support International Inc. All rights reserved. Reproduction or republication strictly prohibited without prior written permission A Vitiligo Support International, Inc. financial statement is available upon written request from the Virginia Office of Consumer Affairs. Mail requests to: Virginia Department of Agriculture and Consumer Services, Office of Consumer Affairs, P.O. Box 1163, Richmond, Virginia 23218. Click here to unsubscribe |
|||||||||||||||||||||||||||

 Now Visit VSI
Now Visit VSI 



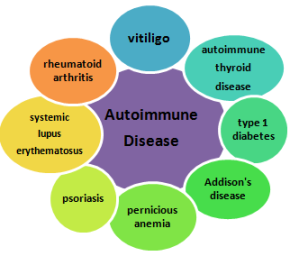 Researchers around the world are investigating the complex interplay of genetics, environmental events, biochemical and immunological events that are believed to be involved in this process of destruction. The process is generally believed to be driven by an autoimmune dysfunction causing the immune system to produce antibodies or cytotoxic T cells, that normally kill virally-infected cells and tumor cells, but instead destroy the body’s own melanocytes. Several other autoimmune disorders occur at increased frequencies with vitiligo, such as autoimmune thyroid disease, Addison’s disease, pernicious anemia, rheumatoid arthritis, psoriasis, type 1 diabetes, and systemic lupus erythematosus, confirming the autoimmune connection discovered by the genetic research of Dr. Richard Spritz and his team at the University of Colorado. Their research has linked several genes found in vitiligo patients to this “family” of autoimmune diseases.
Researchers around the world are investigating the complex interplay of genetics, environmental events, biochemical and immunological events that are believed to be involved in this process of destruction. The process is generally believed to be driven by an autoimmune dysfunction causing the immune system to produce antibodies or cytotoxic T cells, that normally kill virally-infected cells and tumor cells, but instead destroy the body’s own melanocytes. Several other autoimmune disorders occur at increased frequencies with vitiligo, such as autoimmune thyroid disease, Addison’s disease, pernicious anemia, rheumatoid arthritis, psoriasis, type 1 diabetes, and systemic lupus erythematosus, confirming the autoimmune connection discovered by the genetic research of Dr. Richard Spritz and his team at the University of Colorado. Their research has linked several genes found in vitiligo patients to this “family” of autoimmune diseases.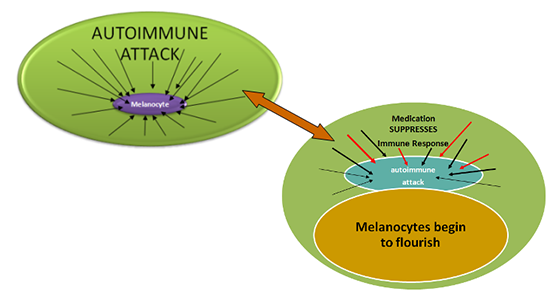
 Therapies approved for treatment of a specific disease are sometimes later found to be effective in treating other diseases as well, either by accident or through scientific evaluation. Repurposing of existing therapies for new disease applications offers a big advantage to both pharmaceutical companies and patients. The costs of adding new disease applications for an already-approved drug are reduced because the relative safety of the drug has already been established. This is especially important for those patients with diseases for which there is limited treatment. In vitiligo, two such drugs that were “repurposed” are tacrolimus (Protopic) and pimecrolimus (Elidel), both of which were originally developed for use in treating eczema before also being used in treating vitiligo.
Therapies approved for treatment of a specific disease are sometimes later found to be effective in treating other diseases as well, either by accident or through scientific evaluation. Repurposing of existing therapies for new disease applications offers a big advantage to both pharmaceutical companies and patients. The costs of adding new disease applications for an already-approved drug are reduced because the relative safety of the drug has already been established. This is especially important for those patients with diseases for which there is limited treatment. In vitiligo, two such drugs that were “repurposed” are tacrolimus (Protopic) and pimecrolimus (Elidel), both of which were originally developed for use in treating eczema before also being used in treating vitiligo. Afamelanotide (brand name Scenesse) is being tested for its ability to accelerate narrowband UVB’s (NB-UVB) ability to repigment vitiligo. Afamelanotide’s photoprotective capacity and ability to promote tanning (stimulate melanin production while protecting the skin from damage associated with light therapy) offer the potential of shortening this repigmentation time. Recently released results of a six-month, three-center, Phase IIa US pilot trial of the drug would seem to confirm this potential benefit.
Afamelanotide (brand name Scenesse) is being tested for its ability to accelerate narrowband UVB’s (NB-UVB) ability to repigment vitiligo. Afamelanotide’s photoprotective capacity and ability to promote tanning (stimulate melanin production while protecting the skin from damage associated with light therapy) offer the potential of shortening this repigmentation time. Recently released results of a six-month, three-center, Phase IIa US pilot trial of the drug would seem to confirm this potential benefit. resulting in repigmentation of vitiligo-affected skin in a few studies. In an effort to prove the efficacy of this form of light in vitiligo, University of British Columbia researchers are recruiting 12 vitiligo patient volunteers to participate in a single-blind, randomized clinical trial in which the effects of high-intensity red light will be compared against a low-intensity red laser. The test is scheduled to be completed by December 2013.
resulting in repigmentation of vitiligo-affected skin in a few studies. In an effort to prove the efficacy of this form of light in vitiligo, University of British Columbia researchers are recruiting 12 vitiligo patient volunteers to participate in a single-blind, randomized clinical trial in which the effects of high-intensity red light will be compared against a low-intensity red laser. The test is scheduled to be completed by December 2013.  A therapy available in Russia has been reported to be effective in halting the progression of vitiligo. At the 2013 European Academy of Dermatology and Venereology (EADV) Symposium held in Cracow, Poland, data was presented on 60 patients whose active non-segmental vitiligo was halted following intramuscular injections of ODHAA. In 44 of the 60 patients (73.3%), the progression of vitiligo was stopped and some repigmentation was observed. After 12 months, their disease remained stable. No side effects were reported and the treatment was well-tolerated.
A therapy available in Russia has been reported to be effective in halting the progression of vitiligo. At the 2013 European Academy of Dermatology and Venereology (EADV) Symposium held in Cracow, Poland, data was presented on 60 patients whose active non-segmental vitiligo was halted following intramuscular injections of ODHAA. In 44 of the 60 patients (73.3%), the progression of vitiligo was stopped and some repigmentation was observed. After 12 months, their disease remained stable. No side effects were reported and the treatment was well-tolerated.  Simvastatin, a common anti-cholesterol medication, is being tested as a vitiligo therapy as part of a Phase II, randomized, placebo-controlled trial at the University of Massachusetts Memorial Medical Center. The first reports of simvastatin having promise in vitiligo were in 2004. A man reported to his doctors that his vitiligo improved after taking high-dose simvastatin to control high blood cholesterol levels. They noted that his vitiligo appeared mild. He then started a clinical trial of a new cholesterol medication, atorvastatin, stopping simvastatin. A couple of months later, the doctors noted that his depigmentation had increased. This depigmentation ultimately regressed after he resumed taking simvastatin.
Simvastatin, a common anti-cholesterol medication, is being tested as a vitiligo therapy as part of a Phase II, randomized, placebo-controlled trial at the University of Massachusetts Memorial Medical Center. The first reports of simvastatin having promise in vitiligo were in 2004. A man reported to his doctors that his vitiligo improved after taking high-dose simvastatin to control high blood cholesterol levels. They noted that his vitiligo appeared mild. He then started a clinical trial of a new cholesterol medication, atorvastatin, stopping simvastatin. A couple of months later, the doctors noted that his depigmentation had increased. This depigmentation ultimately regressed after he resumed taking simvastatin.



 The repigmentation process can begin from the borders of some vitiligo lesions; however, most repigmentation begins as small circular islands around hair follicles which gradually extend outward. Though not a guarantee, generally speaking, having hairs that still have their natural color within the vitiligo lesion is a good sign that there are remaining melanocytes to support repigmentation.
The repigmentation process can begin from the borders of some vitiligo lesions; however, most repigmentation begins as small circular islands around hair follicles which gradually extend outward. Though not a guarantee, generally speaking, having hairs that still have their natural color within the vitiligo lesion is a good sign that there are remaining melanocytes to support repigmentation.


