| Click Here to View VSI Newsletter in Browser |
 |
||||||||||||||||||||||||
This Newsletter Has Been Generously Sponsored By:
|
||||||||||||||||||||||||
|
Dear VSI Members, Friends, and Donors, There is nothing more frustrating than waiting for months to see a dermatologist, only to be told that there are no vitiligo treatments; treatments don’t work; treatments are more dangerous than the disease; or, adding insult to injury, vitiligo won’t hurt you - just go home and be glad you don’t have something more dangerous, like cancer.
No one is MORE frustrated by these thoughts and words than VSI.
Though these problems do still occur, the situation has improved tremendously in the years since VSI began working on a national level to educate all vitiligo stakeholders. VSI has worked tirelessly to provide current and accurate information to vitiligo patients, researchers, clinicians, pharmaceutical companies, insurance companies, national legislators, and a host of other professional organizations. VSI has given presentations on a wide variety of topics, such as barriers to treatment; the importance of vitiligo research funding; breaking down insurance barriers, and, “Life Altering Skin Disease from a Patient’s Perspective.” VSI listens. VSI cares. VSI is working for you. Click here to learn what makes VSI different from other patient groups.
Sincerely,
While researching medical news for this newsletter, I came across a recent clinical trial using piperine and NB-UVB for vitiligo. This is not the first time piperine has been in the news for vitiligo, though it is the first clinical trial with humans I’ve seen. Recalling the furor among VSI members 10 years ago when piperine made its debut, it made me wonder where it had been all these years. What Exactly is Piperine?
As I continued my research, it seemed that most all roads led back to the same person, Amala Soumyanath, B.Pharm., Ph.D. Dr. Soumyanath received both her Pharmacy degree and PhD. in the field of Pharmacognosy (the scientific study of medicinal plants) at King’s College, University of London. Currently, she is an Associate Professor of Neurology in the School of Medicine at Oregon Health & Science University (OHSU), where her interest and research into piperine continue. Between the years 1997-2002, Dr. Soumyanath and her colleagues at King’s College completed multiple studies using different chemical analogs of piperine, testing their ability to stimulate melanocyte proliferation in culture dishes. In time, they narrowed their research to the two most successful compounds, THP and rCHP, and began their research on lightly colored mice.
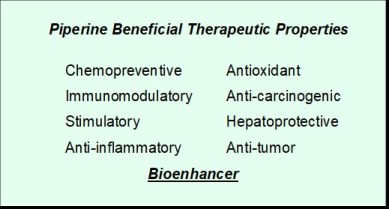
Piperine Research on Mice In a 2008 study, the King’s College research group conducted a study with mice that were randomized to receive topical application of the compounds or only a topical vehicle without the compounds. Some mice received continuous treatment, while some received interrupted treatment in order to evaluate fading and repigmentation. For 4 weeks, all received the topical twice a day, five days a week. Some also received ultraviolet irradiation (UVR) 3 days a week, and some did not receive UVR. They concluded that the group receiving the topical compounds produced greater pigmentation than the vehicle group, with only low levels of inflammation. Those receiving the combo of topical compound and UVR had darker pigmentation and greater numbers of melanocytes than either the compound or UVR alone. They remarked that those in the UVR interruption group had a decrease in pigmentation, but did not have pigment loss. They felt their results in this study supported the potential use of these compounds for treating vitiligo. Funding In 2008, AdPharma, Inc., a clinical trials management and drug development company, announced collaborations with OHSU to advance the compounds through pharmaceutical development. During this time, experiments funded by AdPharma verified that piperine stimulated replication of human melanocytes in culture, as well as within a reconstructed skin model. Of utmost significance, this research seemed to indicate an inhibitory effect on cultured melanoma cells, as well as preventing melanoma cell growth in a reconstructed full skin model. In May 2008, a VSI member contacted AdPharma, requesting information on the status of piperine treatment for vitiligo. He received a reply from Vivekananda Ramana, MD. Executive VP, Clinical Affairs and COO, AdPharma, Inc. stating that they anticipated “conducting clinical trials in patients over the next six months.” Unfortunately, that apparently never took place. NIH Funding Proposal VSI recently corresponded with Dr. Soumyanath who is still very much involved with the piperine research. Though AdPharma is no longer involved, she and her OHSU colleagues obtained funding to study piperine in a mouse model of melanoma. The preliminary results of that research found no evidence of increased melanoma from piperine. With the recent positive clinical trials, and growing research showing that piperine may not only not cause an increased risk of melanoma, but may even prevent or reduce the risk, Dr. Soumyanath conveyed that the time may be right to once again approach the NIH for funding a clinical trial in the US. The success of piperine treatments in vitiligo recently carried out by Iranian researchers, as discussed in the Medical News Updates section of this newsletter, make the potential for a US trial even more exciting. In November of 2016, a different group of researchers from Iran released a thorough and detailed review on the therapeutic and medicinal effects of piperine. They noted the following: “Piperine is the major bio-active component of pepper, which imparts pungency and biting taste to it. This naturally occurring alkaloid has numerous demonstrated health effects and beneficial therapeutic properties.”
Bioenhancers are drug facilitators. They increase the bioavailability and efficacy (therapeutic effectiveness) of a drug with which they are combined. Piperine has been found to increase the bioavailability of different drugs, in a range from 30 to 200%. The benefits of adding a bioenhancer include the ability to reduce the drug dosage, resulting in reduced cost. This will not only make some very expensive treatments more affordable (such as anti-cancer drugs), but can also reduce the amount of raw materials, which in some cases are quite rare. However, there can also be a downside to these bioenhancers, as if the dosage of the combined drug is not reduced, it may increase the risk of serious side effects. How the Body Reacts to Bioenhancers The body has built in mechanisms for controlling the exposure of its cells to nutritional and other substances. Piperine’s properties enable it to manipulate these mechanisms by inhibiting a number of the enzymes responsible for metabolizing drugs and nutritional substances, allowing more of the substances to enter the body in active form. The difficulty, and potential danger, comes from the many different ways the body processes different drugs/substances, making reactions both complex and unpredictable. In some instances, piperine inhibits enzymes, resulting in increased drug absorption. However, in other cases, it increases the body’s production of enzymes, resulting in decreased absorption, which can be equally dangerous. Many supplements that include a popular piperine supplement also contain some form of turmeric/curcumin, which opens an entirely new category of side effects and warnings and can be especially dangerous for anyone taking a blood thinner. While several major categories of drugs have been found to increase absorption when combined with piperine, researchers are quick to note that this list is constantly changing. The list below is for reference only and not to be considered complete.
Piperine in the USA The topical analog compounds being used in research have not received FDA approval for human use in the United States. However, piperine and associated black pepper products are available as over-the-counter dietary supplements in a multitude of products, such as extracts, essential oils, aromatherapy, powder, and capsules. Over-the-Counter Drugs (OTC) After reading about the possibilities of treating vitiligo with piperine, it may be tempting to try one of the above supplements and begin self-treating. However, it’s important to understand that this is not without risk, and could have very dangerous consequences. Many people are under the incorrect assumption that vitamins and supplements are natural and therefore safe to consume or use without precaution. Many also assume that the Food and Drug Administration (FDA) monitors OTC products the same way as prescribed drugs, but this assumption also is not true. A 2013 study found that more than half of FDA Class 1 drug recalls between 2004 and 2012 were for dietary supplements. Class 1 recalls are reserved for drugs or supplements for which there is a “reasonable probability that [their use] will cause serious adverse health consequences or death.” A 2013 research letter published in the JAMA Internal Medicine (a peer-reviewed medical journal published by the American Medical Association), reporting on OTC and compounded vitamin D, found pills ranging from as low as 9 percent to as high as 146 percent of the stated dose.
The bottom line is that, as with other OTC medications, piperine and black pepper derivatives are not regulated for content, labeling, or dosage. Research has shown that these substances can have very powerful and dangerous side effects in very small doses. You have no way of knowing if an essential oil, powder, or pill containing piperine that you’re buying is the strength it says on the label, far less, or far greater. You should always consult your physician or healthcare provider before taking any dietary supplement. Though it seems we are much closer to having topical piperine clinical trials in the USA, we will have to wait until safety and efficacy have been established and approved.
Calling all Medicare Vitiligo Patients!
Part of the problem is that currently there is no accepted standard for coverage of vitiligo treatments. Each company has their own policies and guidelines, many times based on outdated and incorrect information. What most people may not realize is just how much private insurance coverage can be affected by Medicare policies. Over 55 million Americans are now covered by Medicare or Medicare Advantage, making it the nation’s largest health insurance program. Consequently, many, if not most, insurance providers establish their baseline standards and procedures to follow the Medicare model. Medicare has agreed to meet with us – but requires that we bring Medicare letters of denial for a vitiligo treatment. If you are reading this newsletter and you, or someone you know, was denied coverage for a vitiligo treatment by Medicare, please contact VSI immediately. You could help make a difference for future insurance coverage for ALL vitiligo patients! Click Here to Contact VSI
What's On Your Mind? Q. Can chemicals cause vitiligo to develop many years after contact?
Highlights of recently-published medical Topical Piperine Combined with NB-UVB for Vitiligo Researchers in Tehran, Iran released a study in May 2018 on the effect of topical piperine combined with NB-UVB as a treatment for vitiligo. They knew from long-standing research that piperine stimulates melanocyte (pigment-making cells) proliferation. Though there have been research studies with topical piperine in vitro and animal models, human research has been limited. This double-blind clinical trial compared the results between two groups of patients with facial vitiligo. Both groups received standard NB-UVB phototherapy every other day for three months. The “case” group was treated with a topical piperine solution prepared by dissolving 95% pure piperine in a solvent mixture [dimethyl sulfoxide: isopropyl alcohol and glycerol] (20: 80). The “control” group received a topical placebo with the same formulation minus the piperine. In the case group, 10 patients experienced a mild burning sensation on the skin, and 6 patients experienced redness on the treated area. These side effects were temporary and were cleared by using zinc oxide ointment. The percentage of repigmentation was assessed in both groups after 1, 2, and 3 months. The difference in repigmentation between the first and third months was described as remarkable.
The researchers noted that because the piperine group experienced such accelerated improvement, another benefit of piperine would be reduction in overall UVB exposure. Evaluation of Serum Zinc Level in Patients with Vitiligo Researchers from Tehran, Iran recently completed a case-control study comparing the serum zinc level from those with vitiligo to a non-vitiligo control group. The study included 103 vitiligo patients with no other dermatologic or systemic diseases and no zinc supplementation over the previous 4 weeks, and 103 sex and age-matched individuals without vitiligo.The vitiligo group was divided into three subtypes: Research has shown a destructive relationship between oxidative stress and melanocyte apoptosis, leading to the development of vitiligo. Because zinc is known for its ability to eliminate free radicals, its role in producing melanin, and its anti-apoptotic properties, the authors conducted the study to evaluate and compare the serum zinc level of those with vitiligo to those without.
Using atomic absorption spectrophotometry to measure the serum zinc levels, researchers found the levels to be nearly the same for the control group and the focal vitiligo group. However, those in the generalized vitiligo group had significantly lower zinc levels. The number of participants with mucosal vitiligo was too low for comparison.
The researchers also noted a correlation between longer disease duration (length of time since disease onset) and lower zinc levels. In other words, the longer a person has had vitiligo, the greater their risk for developing reduced zinc levels.
New Paid Study in Lynchburg VA!
Earn Funding for VSI with Amazon and eBay Online Shopping Can Benefit VSI!
Support VSI Through Ebay
|
|||||||||||||||||||||||
| Copyright © 2018 Vitiligo Support International Inc. All rights reserved. Reproduction or republication strictly prohibited without prior written permission A Vitiligo Support International, Inc. financial statement is available upon written request from the Virginia Office of Consumer Affairs. Mail requests to: Virginia Department of Agriculture and Consumer Services, Office of Consumer Affairs, P.O. Box 1163, Richmond, Virginia 23218. Unsubscribe from the original email |
||||||||||||||||||||||||