In This Issue
View Past Newsletters
VSI Medical and Scientific Advisory Committee
Committee Chair:
Pearl E. Grimes, M.D.
Ted A. Grossbart, Ph.D.
Sancy A. Leachman, M.D.
I. Caroline Le Poole, Ph.D.
Mauro Picardo, M.D.
Nanette B. Silverberg, M.D.
Richard A. Spritz, M.D.
Alain Taieb, M.D., Ph.D.
Wiete Westerhof, M.D., Ph.D
Message from the Executive Director
 Dear VSI Members, Friends, and Donors,
Dear VSI Members, Friends, and Donors,
From April 21 – 23, 2022, I was honored to attend the 3rd
VIPOC (Vitiligo International Patient Organizations Conference)
in Amsterdam, Netherlands.
Vitiligo patient advocacy organizations from 20 nations joined together with researchers, physicians, and pharmaceutical and industry representatives, to strengthen collaborations and increase vitiligo awareness.

VIPOC Board:
From Left Jean-Marie Meurant France, Maya Tulpule India, Georg Pliszewski Germany, Jackie Gardner USA, Nicolle Maquignon France, Stephen Taylor USA, Sharon King USA, Catherine Davidson UK
Presentations and workshops focused on topics such as quality-of-life impact, emotional
and psychological burden, cultural and social impacts, and the need for increased
resources for children to prevent bullying and isolation.
Participants were given the opportunity to share their thoughts directly with
pharmaceutical representatives regarding the need for improved and affordable treatments with fewer side-effects, and greater collaboration and inclusion of patient input in treatment research, discussions, and decisions.
Because VSI members so graciously shared their most heartfelt thoughts and
experiences in our Spring newsletter survey (My Vitiligo Story), my presentation
centered on these survey results, leaving no question that vitiligo is a life-altering
skin disease, with very deep-seated emotional and psychological effects impacting
life’s decisions every single day.
We received many poignant comments that rang so true to me:
“Every single patient is different. We are not a monolith. It’s annoying to be expected to have the same emotional reaction or mindset as everyone else (e.g. “embrace it” because a famous model has it and is thriving).”
“Just because it is not physically painful does not mean it doesn’t hurt.”
“My family has always said, “Oh, we don’t even notice it.”
This makes me feel dismissed because it has been devastating for me to have vitiligo.
Nearly 70% reported:
Not feeling their own family understands what it is like to have vitiligo
Nearly 75% Reported:
Feeling their life would have been better if they didn’t have vitiligo
Nearly 85% Reported:
Experiencing feelings of sadness or depression
Support VSI
As a 501(c)(3) nonprofit organization,
VSI is dependent on public funding to operate.
If it’s Important to You AND That VSI Continues its Efforts on Your Behalf to:
- Advocate and Increase Vitiligo Awareness
- Make Sure the Voice of the Vitiligo Patient is Heard and Understood
- Provide Trusted, Current Resources like these Newsletters
Please Consider a Donation to Sustain this Vital Work.
Together We Can Make a Difference!
Sincerely,
Jackie Gardner
Executive Director

VSI is proud to announce that we have been awarded the
GuideStar Gold Seal of Transparency for 2022!
With a database of more than 1.8 million IRS-recognized organizations,
GuideStar is the most complete source of information about U.S. charities.
Search Vitiligo on Guidestar
COVID-19 and Vitiligo:
What We Know Now
When COVID-19 vaccines first became available, VSI was bombarded with questions regarding the risks or benefits, with many worrying that the vaccine would be a trigger causing their vitiligo to spread.
With all available scientific and medical resources being dedicated to saving the lives of those affected by one of the world’s deadliest pandemics, other than expert speculation, there was very little information available on topics related to impact on specific diseases such as vitiligo.
VSI’s Spring 2021 Newsletter was devoted to providing answers to as many of the questions and concerns for which we could find answers. The problem was that reputable sources were limited and the available information was very fluid with constant updates and changes, as well as conflicting reports.
There was additional concern regarding the safety of the vaccines for those with autoimmune (A/I) diseases. Part of that confusion stemmed from mixed messaging indicating that those with A/I diseases had been excluded from the vaccine clinical trials, which not only was inaccurate, but very misleading.
When you mix all of that with the long-held misconception of many that having an autoimmune disease indicates a person has a weakened immune system that makes them immunocompromised, the ensuing anxieties and questions of safety continued to grow.
Where are We Now?

With nearly a year-and-a-half of real-world experience in the history books, disease-specific research is beginning to trickle in. We thought it would be a good time to take a closer look at the data and begin replacing some of the myths and speculation with factual evidence.
We will begin by reviewing some of the misconceptions, such as the theory that having an autoimmune disease means you have a weakened or immunocompromised immune system.
Immunocompromised:
When an immune system is weaker than it should
be, compromising its ability to fight off infections.
With autoimmunity, rather than being weakened, part of the body’s immune system is
actually overactive, and is mistakenly attacking part of itself. With vitiligo, the immune
system is attacking the body’s pigment-making cells.
Dr. Bernard Khor of the Benaroya Research Institute Explains:

“Immune dysregulated could be a better term for most people with
autoimmune disease. That means the immune system isn’t quite
working right, but that doesn’t necessarily impact your ability to
fight off infection or respond to a vaccine.”
Some people with some autoimmune diseases (such as rheumatoid arthritis) take systemic
immunosuppressing medications to intentionally reduce their immune system’s attack on their body. These individuals were excluded from the clinical trials, due to customary clinical trial protocol.
COVID-19 vaccine clinical trials did NOT exclude
People with A/I conditions like vitiligo that are manageable
without taking systemic immunosuppressant drugs
COVID-19 vaccine clinical trials DID exclude:
Individuals taking systemic immunosuppressant drugs.
Similar to the confusion surrounding the autoimmune exclusion, many assumed that those taking immunosuppressing drugs (aka immunosuppressed individuals) were excluded from the clinical trials due to safety concerns. However, not only is that not true, but the exact opposite is actually the case.
Those with A/I conditions who were on immunosuppressant drugs were excluded from the clinical trials because these drugs, as noted in their name, suppress the immune response and can reduce “the body’s ability to mount a robust immune response to a COVID-19 vaccine.” The clinical trials at that stage were trying to determine how safe and effective the vaccines were for the general population. They wouldn’t include a specific group known to have a reduced immune response.
Immunosuppression, COVID-19, and the Vaccines:
February 2022 research study carried out
at the Univ. of Michigan confirmed the following:
- Immunosuppressed individuals (I/I) are at a higher risk of SARS-CoV-2 infection and COVID-19 hospitalization.
- Receiving one of the three approved COVID-19 vaccines substantially lowered this risk.
- Reduced immune response resulted in lower vaccine effectiveness and in immunity waning more quickly for I/I than the general population.
- Booster doses were highly effective at reducing risk of infection and hospitalization for these individuals.
- Receiving vaccine booster doses is critically important for I/I.
According to the above study, the C-19 vaccines have been shown to be safe, effective, and critically important to the immunocompromised population.

A Closer Look at Vaccine Response
in the Autoimmune Population
Now, with over 12 billion vaccines having been administered worldwide, more disease-specific research is beginning to become available. We can take a look at the safety, efficacy and side effects of some of the different vaccines on some of the sub-population groups.
A group of researchers in Amsterdam, Netherlands decided to do just that. Noting an existing problem of vaccine hesitancy in general among patients with autoimmune diseases, and with the hope of dispelling any myths that might stand in the way of these individuals getting a C-19 vaccine, these investigators felt it important to take a closer look into the tolerability and any adverse effects from the different types of C-19 vaccines among this group.
They collected data from 505 patients with systemic A/I diseases
and 203 healthy controls (without A/I diseases).
Receiving *AstraZeneca Vaccine:
231 A/I patients (46% of that group)
104 Healthy controls (51%)
Receiving Pfizer Vaccine:
209 A/I patients (41%)
90 healthy controls (44%) controls
Receiving Moderna Vaccine:
65 A/I patients (13%)
9 healthy controls (4%)
*AstraZeneca vaccine is not
approved for use in the USA.
Adverse Events Were Reported by the Following
Mild: Unpleasant reactions that did not limit daily activities
- 258 A/I patients (51%)
- 106 health controls (52%)
Moderate: Reactions limiting daily activities
- 105 A/I patients (21%)
- 38 controls (19%)
Severe: Reactions requiring medical attention
- 6 A/I patients (1%)
- 0 healthy controls
Serious: Reactions resulting in hospital admission
- No serious reactions were reported
Systemic reactions were defined as: Fever, chills, general malaise,
nausea, diarrhea, muscle aches, joint pain, headache, and fatigue.
The most frequently reported systemic events were fatigue and headache.
222 A/I patients (44%)
82 healthy controls (40%)
The most commonly reported adverse event was pain at the injection site.
This was consistent across all vaccines.
196 A/I patients (39%)
82 healthy controls (40%)
Their results indicated a perceived overall improvement in the quality of life,
with 75% of the A/I patients and 76% of the controls reporting that they felt safer
within 2 weeks after their first C-19 vaccine.
Did the Vaccines Increase Disease Activity?
All patients with A/I diseases were asked if they experienced any changes in their disease
within two months* after a C-19 vaccination. The study authors noted that only a very small proportion (5%), reported a worsening of their autoimmune disease up to 2 months
after receiving the vaccine.
Their overall data indicated that the C-19 vaccines were well tolerated
and adverse events were comparable to the control group,
(regardless of the type of vaccine).
They hope their findings will be reassuring
and reduce vaccine hesitancy in this group.
The above study did not disclose which specific autoimmune diseases were
involved in the study, and only allowed respondents 2 months before assessing
increase in disease activity.
*While it is possible for vitiligo pigment loss to become evident within 2 months after a trigger, it is not uncommon to take 3 months or longer to become visibly apparent.
In an effort to provide additional information more specific to vitiligo, below we present
findings from another similar study in Bucharest, Romania that allowed a 6-month follow-up period after receiving a COVID-19 vaccine to assess disease flares, and this study
specifically cited the inclusion of vitiligo patients.
“COVID-19 Vaccine Does Not Increase the Risk of Disease Flare-Ups
Among Patients with Autoimmune and Immune-Mediated Diseases.”
This study, included a total of 623 participants, with 416 having been vaccinated and
207 non-vaccinated, which served as the control group in terms of flare-ups.
COVID-19 Vaccines Administered
Comirnaty (BioNTech/Pfizer, Germany, Mainz) (86%)
Vaxzevria (Oxford/AstraZeneca, Nijmegen, Netherlands) (9%)
Spikevax (Moderna Biotech, Cambridge, USA) (3%)
Janssen vaccine (Johnson & Johnson, Leiden, Netherlands) (2%)
Top symptoms after receiving a vaccine:
Pain at injection site, fatigue, headache, chills, joint pain, and myalgia.
During the 6 month follow-up period, a total of 42 flare-ups were reported.
25 (6%) in the vaccinated group
17 (8%) in the non-vaccinated group
In terms of flare-up development, they reported no significant difference
between the different types of COVID-19 vaccines administrated.
They also found no statistical difference concerning the risk of flare-ups
between vaccinated and non-vaccinated patients.
There was only 1 report of a disease flare-up among those
with cutaneous diseases, which included participants with vitiligo.
The greatest number of flare-ups were associated with those having more than one immune-mediated disease (38%), and those who experienced a flare-up in the previous year (73%).
Similar to the previous study, the study authors concluded:
“There should not be an obvious reason for
vaccine hesitancy among this category of patients.”
COVID-19 and Vaccine Considerations
For those who might be avoiding a C-19 vaccine or booster due to concerns that it might make their vitiligo spread, other factors to consider:
If I get C-19, how sick will I get?
If I get C-19, will it cause my vitiligo to spread?
If I get C-19, are there others in my household that I might infect?
The bottom line is that you and your medical provider are in the
best position to know what is best for you and your health.
Calling all Medicare Vitiligo Patients!
 VSI regularly hears from patients from all over the United States who have been denied insurance coverage for their vitiligo treatment. VSI has been fighting this battle on a national level for several years. While we are definitely making progress, it’s been a slow road.
VSI regularly hears from patients from all over the United States who have been denied insurance coverage for their vitiligo treatment. VSI has been fighting this battle on a national level for several years. While we are definitely making progress, it’s been a slow road.
Part of the problem is that currently there is no accepted standard for coverage of vitiligo treatments. Each company has their own policies and guidelines, many times based on outdated and incorrect information.
What most people may not realize is just how much private insurance coverage can be affected by Medicare policies. Over 55 million Americans are now covered by Medicare or Medicare Advantage, making it the nation’s largest health insurance program. Consequently, many, if not most, insurance providers establish their baseline standards and procedures to follow the Medicare model.
Medicare has agreed to meet with us – but requires that we bring Medicare letters of denial for a vitiligo treatment.
If you are reading this newsletter and you, or someone you know, was denied coverage for a vitiligo treatment by Medicare, we really need a copy of your Medicare Denial letter. You could help make a difference for future insurance coverage for ALL vitiligo patients!
Click Here to Contact VSI
What's On Your Mind?
Q. Which is a better treatment, excimer laser or NB-UVB?
I am searching for a doctor who is able to prescribe light treatments for me, but I’m trying to understand the difference between narrow band UVB and excimer laser. Is one better than the other?
A. There are various factors to consider to determine the best choice.
 Excimer laser uses a wavelength similar to NB-UVB, but delivers a targeted, narrow beam. It is very effective, but because of the size of the beam it is most often used to treat very small areas of depigmentation. Some doctors will begin with laser therapy for patients who have only a couple of very small areas of depigmentation. However, laser is more typically used for very small spots remaining after NB-UVB treatment.
Excimer laser uses a wavelength similar to NB-UVB, but delivers a targeted, narrow beam. It is very effective, but because of the size of the beam it is most often used to treat very small areas of depigmentation. Some doctors will begin with laser therapy for patients who have only a couple of very small areas of depigmentation. However, laser is more typically used for very small spots remaining after NB-UVB treatment.
 NB-UVB phototherapy is now the gold standard light treatment for vitiligo, and the initial treatment of choice for patients with vitiligo involving greater than 10 percent of the body surface area (BSA). A larger NB-UVB light unit reduces treatment time by treating larger areas in a single exposure. It can also catch and treat areas in the process of depigmenting before they become noticeable.
NB-UVB phototherapy is now the gold standard light treatment for vitiligo, and the initial treatment of choice for patients with vitiligo involving greater than 10 percent of the body surface area (BSA). A larger NB-UVB light unit reduces treatment time by treating larger areas in a single exposure. It can also catch and treat areas in the process of depigmenting before they become noticeable.
Another bonus of NB-UVB therapy is that when used on the full body, research has shown it to be effective at stabilizing active vitiligo. So, not only are you regaining pigment, but you may also be halting disease progression in other areas.
Medical News Updates
Highlights of recently-published medical
articles on vitiligo and its treatments
Can Vitiligo Protect You
From COVID-19?
A group of researchers in the Netherlands is currently involved in a study to learn whether there is a relationship between autoimmune vitiligo and COVID-19. The study authors have written a scientific hypothesis letter outlining their thoughts, based on known risk and protective factors involved with both vitiligo and the potential severity and mortality of COVID-19.
We know from research that non-segmental vitiligo (NSV) is an autoimmune-mediated inflammatory disease, driven by genes involved in immune activation and regulation. It is thought that these altered vitiligo-associated genes that play a role in predisposing patients to autoimmunity and vitiligo may also increase their antiviral immunity.
Innate Antiviral Immunity
An antiviral is something that kills a virus and/or suppresses its ability to replicate.
Innate antiviral immunity is something you’re born with, not something that you develop from virus exposure or vaccine. Intrinsic antiviral immunity is one kind of innate antiviral immunity that stops a viral attack directly by stopping the cells from allowing it entry.
Coronavirus Background:
SARS-CoV-2 is a virus and an infection.
COVID-19 is the respiratory disease caused by the SARS-CoV-2 virus.
The first human coronavirus was identified in 1965, causing the common cold. Since that time, the coronavirus has become a large family of viruses known to cause upper respiratory tract illnesses in humans.
SARS (Severe Acute Respiratory Syndrome) was first discovered in Asia in February 2003. In 2019, a new strain was discovered and in February 2020, named “severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2)” because, although different, it is genetically related to the virus responsible for the SARS outbreak of 2003. This new strain was identified as the cause/trigger of the coronavirus disease 2019 (COVID-19) outbreak, which in March 2020 was labeled a pandemic by the World Health Organization.
The Study Hypotheses
The study authors hypothesize that because of the activated type 1 immune status and association with genetic antiviral genes of those with NSV, that if they are infected by the SARS-CoV-2 infection, that they would clear the viral infection more efficiently (with less severity) and have a lower risk of going on to develop COVID-19.
However, they also hypothesize that if a person with NSV does go on to develop COVID-19, the associated elevated vitiligo autoimmunity could also potentially trigger the “cytokine- storm” associated with COVID-19 severity and death.
What is a Cytokine?
According to the American Cancer Society, cytokines are small proteins that are crucial in controlling the growth and activity of other immune system cells and blood cells. When released, they signal the immune system to do its job.
What is a Cytokine Storm?
A cytokine storm has been described as a hyperactive immune response. An example might be blowing up your entire house because you have ants in the kitchen.
The problem is two-fold, beginning with an over-active response. Then it’s made worse because the response doesn’t know when to stop and just keeps growing like an out-of-control fire.
The authors hypothesize that due to the cytokines identified as playing a pivotal role in the development of autoimmune diseases, immune activation during the SARS-CoV-2 infection or COVID-19 disease could potentially increase vitiligo disease activity.
The authors will determine disease activity by asking if the vitiligo patients developed any new vitiligo lesions during the 3 months after contracting COVID-19. They were careful to add that any potential effect on disease activity could either be direct, due to the increased immune activation. Or, an indirect response due to the emotional stress experienced during COVID-19 illness.
Testing the Hypotheses
The authors have developed a questionnaire that is currently being distributed worldwide to a large number of NSV patients between the ages of 16 and 55 years.
The Study Authors Have Given VSI Members The Opportunity
To Participate In This Intriguing Research!
There has been so little research regarding the impact of COVID-19 on those with vitiligo.
This is your opportunity to participate in one of the FIRST research studies specifically about COVID-19 and Vitiligo.
To Take the Questionnaire Now: Click Here!
VSI will report the study results once they have been published.

Ruxolitinib Cream for Vitiligo!
FDA Target Date July 18!
We’ve all been anxiously awaiting the FDA approval of the topical JAK inhibitor ruxolitinib cream 1.5% (Opzelura™) which will set precedent as the first ever FDA-approved, “on label” prescription for repigmentation of vitiligo.
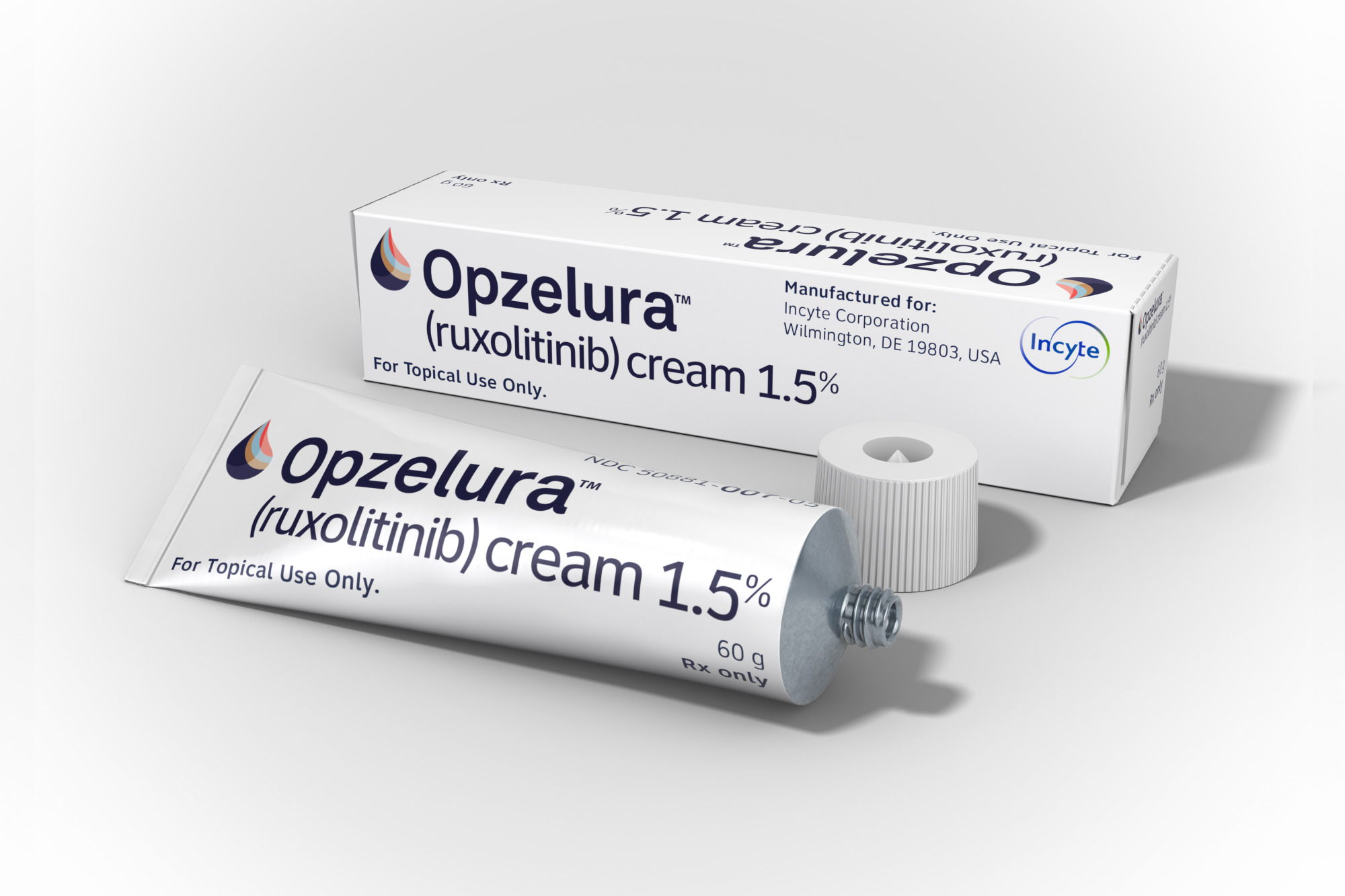 If you’ve been following this news as closely as VSI, you know that the target action date for approval had been set for April 18, 2022. However, on March 14, 2022, the FDA announced they had extended the review period by three months to July 18, 2022.
If you’ve been following this news as closely as VSI, you know that the target action date for approval had been set for April 18, 2022. However, on March 14, 2022, the FDA announced they had extended the review period by three months to July 18, 2022.
As of the date this newsletter went to editing, the FDA July 18 date was still holding. Hopefully by the time you receive your newsletter, Opzelura™ will have been approved for use with vitiligo!
Stay Tuned!
Research & Clinical Trials
Participate in One of the FIRST Research Studies
Specifically About COVID-19 and Vitiligo
Does Autoimmune Vitiligo
Protect Against COVID‐19 Disease?
The Netherlands Institute for Pigment Disorders of the Amsterdam University Medical Centre, is conducting a research to study the influence of vitiligo on COVID-19.
They would like to determine whether having vitiligo might make one more susceptible, or less susceptible to developing COVID-19; how severe the illness is for those with vitiligo that DO develop COVID-19, and what, if any, effect COVID-19 might have on vitiligo disease activity.
They are seeking Participants WITH Vitiligo
As well as
Participants WITHOUT Vitiligo (as controls).
(If you have a friend or family member without vitiligo who would be willing to take the control survey, that would help them tremendously!)
The questionnaire takes 10-15 minutes to complete. ALL data will be confidential and processed anonymously.
———————-
If you DO HAVE Vitiligo: Click here
If you DO NOT HAVE Vitiligo: Click here
New Paid Vitiligo Study
in New York City
Vitiligo Patients Needed
For PAID Research Study at Mt. Sinai
Visits May Occur at one of the Following
Mt. Sinai Medical Office locations:
5 East 98 Street NYC– 5th Floor
234 East 85 Street NYC – 5th Floor
The Mt. Sinai Department of Dermatology Clinical Trials Center is looking for patients diagnosed with vitiligo to participate in a clinical research study.
- If you or your child have been diagnosed with vitiligo
you may qualify to participate. - All ages accepted.
- Only one visit required, which will include a blood draw
and tape strips which are used instead of needle biopsy.
For Additional Information
Contact Dermatology Clinical Trials at:
212-241-3288
(reference biopsy study)
Multi-Center Vitiligo Treatment Clinical Trial
New Locations Added!
Phase 2 Clinical Trial Evaluating
AMG 714 Treatment for Vitiligo
New Study Locations!
California: Irvine and Davis
Connecticut: New Haven
Massachusetts: Boston and Worcester
Michigan: Detroit
New York: Lake Success
As VSI has previously reported, many times when a person experiences a vitiligo relapse, they lose pigment in the exact same location as a previous lesion, due to the discovery of autoimmune memory T cells. This new therapy targets and blocks the memory signaling and also removes these memory T cells, which is showing long-term improvement from short-term therapy.
Participants will be randomized into one of two groups
for a 24 week, double blind period.
Each group will receive 300 mg of AMG 714, or, placebo for AMG 714,
subcutaneously on Day 0 and every 2 weeks thereafter through week 10
(for a total of 6 doses).
At week 24, all participants who have not shown at least 25%
improvement, will begin NB-UVB phototherapy 3 times per week
for a total of 24 weeks.
Key Inclusion Criteria:
- Clinical diagnosis of active or stable non-segmental vitiligo
- At least 0.25% facial depigmentation
- At least 3% depigmentation on the body
- Must be willing to undergo narrow band ultraviolet B (nbUVB) phototherapy
- Must discontinue all vitiligo treatments from screening through the final safety follow-up visit at week 48
Key Exclusion Criteria:
- More than 33% pigment loss on the face or on the total body
- Previous or current diagnosis of other cancer
- May never have used any depigmentation treatments
For Additional Information, Study Locations, and Contact:
CLICK HERE!
Earn Funding for VSI with Amazon and eBay
Online Shopping Can Benefit VSI!
With thousands of items in addition to books, Amazon.com is a one stop-shopping center! Simply shop through the above link (bookmark it for easy reference), and VSI will earn fees, based on a percentage of the sale. The more items purchased by members, the higher the percentage!
Tobi Cares Donation Program
Tobi is an online women’s clothing design label that brings LA style from its design studio direct to its online clothing store at tobi.com. Go to Tobi Cares and sign up for VSI to receive 1% of your purchase amount.
IGIVE SHOPPING
iGive.com offers access to free shipping deals and exclusive coupons, on top of the great deals you’ll find every day through its network of 1,000+ stores, including Pottery Barn, REI, Staples, Petco, Expedia, Best Buy, QVC and many more. Best of all, up to 26% of your purchase at each store is donated to VSI at no cost to you! Let friends and family know so they can support VSI, too. Click register for iGive to get started today, and download the iGive button to automatically benefit VSI whenever you go to an included store. You can also raise a penny per search through iGive’s search engine, isearchigive.com.
GOODSEARCH SHOPPING AND DINING!
GoodSearch and Goodshop, like iGive, offers coupons, discounts, and donations to VSI through its network of over 5,000 stores. Just click the link above to get the savings started and the donations flowing!
* Vitiligo Support International Inc. (VSI) is a participant in the Amazon Services LLC Associates Program, an affiliate advertising program designed to provide a means for sites to earn advertising fees by advertising and linking to Amazon.com.
Earn Funding for VSI with eBay
Support VSI with Ebay!
Do you sell items on eBay, or want to hold an online garage sale? If so, consider donating a percentage of your proceeds to VSI through eBay’s Giving Works program! It’s easy – when you list an item through a Giving Works listing, choose to send 10-100% of the final sale price to VSI. Your listing will have a special placement and designation. You’ll receive a proportional fee credit from eBay, and will also receive a tax donation receipt when the donation is deducted from your funds received. It’s a great opportunity to make some money for yourself and for VSI, so clean out those closets and garages and get selling!Copyright © 2022 Vitiligo Support International Inc. All rights reserved.
Reproduction or republication strictly prohibited without prior written permission.
A Vitiligo Support International, Inc. financial statement is available upon written request from the Virginia Office of Consumer Affairs. Mail requests to: Virginia Department of Agriculture and Consumer Services, Office of Consumer Affairs, P.O. Box 1163, Richmond, Virginia 23218.


